Research Interests
Twenty first century has witnessed a renaissance in the chemistry of main group elements. Activation of inert chemical bonds, an area which was mainly confined to transition metals, can now be performed by reactive main group compounds. Small molecules like H , CO , C H and NH have been activated under mild conditions using main group compounds, thus proving that they can compete with transition metals. While transition metal compounds usually require main group Lewis acids as co-catalysts for bond activation processes, main group compounds themselves exhibit reactivity without the involvement of an additive. We are interested in the isolation of electrophilic main group compounds and employ them as Lewis acid catalysts. Recent contributions from our group in this area are presented below:
2
2
2
4
3
Alkyls, Amides and Hydrides of Group 2 and 12 metals
Our research endeavors are centered on delving into the chemistry of alkali, alkaline earth metals, and Group 12 elements. Our primary focus lies in synthesizing compounds with lower oxidation states and developing stoichiometric reagents and catalysts that hold significance in various organic transformations. Notably, cationic compounds display distinctive structural characteristics and reactivity patterns. However, gaining synthetic access to these cationic species poses challenges due to the dynamic Schlenk equilibria occurring in solution. Our current investigations focus on cationic compounds featuring alkyl, amide, and hydride ligands.


Group 13-15 Lewis Acids
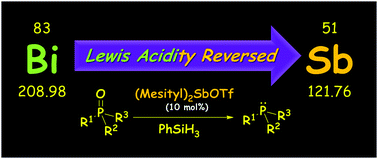
Exploring the properties of main group cations has piqued our interest due to their potential as strong Lewis acids. However, delving into cation chemistry presents certain challenges. One key challenge involves selecting appropriate ligands to stabilize the coordination structure of the dication while maintaining its reactivity. Although cationic chemistry is an evolving field, it has mostly centered around the lighter p-block elements. In contrast, investigating reactive dications among the heavier main group elements remains limited due to the complexities associated with their synthesis.
Recent breakthroughs in understanding the reactivity of aluminum, tin, antimony, and bismuth cations have motivated us to explore their potential as catalysts in the hydrosilylation of unsaturated chemical bonds. These findings hint at promising applications of these cations in catalytic transformations with important implications for various chemical processes.